FDA Hormone Therapy Warning Changes: What the Evidence Actually Shows
🎧 Audio Overview
summary, or read the transcript below:
View Audio Transcript
The FDA is planning to revise its black box warnings on menopausal hormone therapy—warnings that have shaped prescribing patterns for over two decades. This reflects accumulated evidence that hormone therapy’s risks and benefits depend critically on timing and age.
The original 2002 Women’s Health Initiative study found increased risks of breast cancer and heart disease. But the study enrolled women with an average age of 63—many over a decade past menopause. That timing matters profoundly.
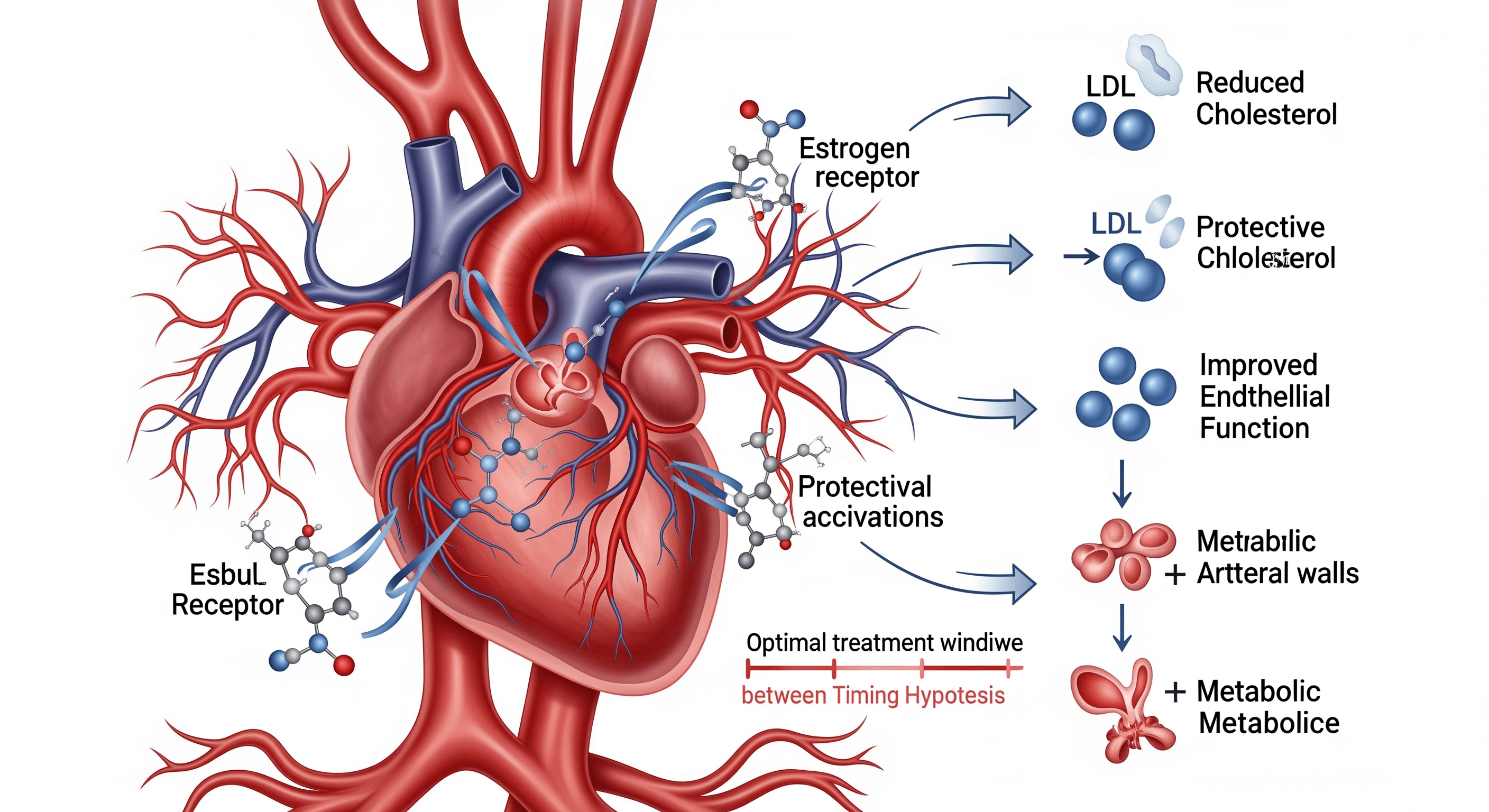
What emerged from deeper analysis is the “timing hypothesis.” Evidence shows that hormone therapy initiated within 10 years of menopause onset—or in women under age 60—demonstrates a different safety profile than therapy started in older women.
Younger women starting hormone therapy closer to menopause show reduced cardiovascular events, lower all-cause mortality, improved metabolic markers, and maintained bone density. A 2015 Cochrane review found that hormone therapy started before age 60 was associated with reduced cardiovascular and all-cause mortality.
The mechanism involves vascular health. Before significant atherosclerotic plaque develops, estrogen appears protective—improving endothelial function and reducing LDL cholesterol. But once substantial plaque exists, estrogen may destabilize it.
Breast cancer risk depends heavily on whether you’re using estrogen alone or combined with progestin. Different progestins carry different risks, with bioidentical progesterone possibly safer than synthetic versions.
The anticipated FDA changes don’t mean hormone therapy is risk-free. They mean we’ve learned that risks and benefits vary substantially based on patient characteristics and treatment timing. For women in their 50s experiencing significant menopausal symptoms, evidence suggests short-to-moderate term hormone therapy carries a favorable risk-benefit profile.
The FDA is planning to revise its black box warnings on menopausal hormone therapy—warnings that have shaped prescribing patterns and patient decisions for over two decades. This potential policy shift reflects accumulating evidence that hormone therapy’s risks and benefits depend critically on timing, age, and patient selection.
For women navigating menopause and weighing treatment options, understanding what the evidence actually shows—beyond the headlines that have dominated since 2002—matters significantly.
The Original Warning and Its Impact
In 2002, the Women’s Health Initiative study was halted early after finding increased risks of breast cancer, heart disease, stroke, and blood clots among women taking combined estrogen-progestin hormone therapy. The findings triggered immediate changes: the FDA mandated black box warnings, prescriptions plummeted, and hormone therapy became something many physicians avoided prescribing and many women feared taking.
That initial interpretation, while understandable given the study design, missed critical nuances that subsequent analyses revealed. The WHI enrolled women with an average age of 63—many over a decade past menopause onset. These weren’t women seeking treatment for acute menopausal symptoms but older women being given hormones to test whether they might prevent chronic disease.
That timing matters profoundly. As reanalysis of the WHI data and subsequent studies have shown, hormone therapy’s risk-benefit profile differs dramatically based on when treatment begins.
The Timing Hypothesis
What emerged from deeper analysis of the WHI and other studies is often called the “timing hypothesis” or “window of opportunity.” The evidence suggests that hormone therapy initiated within 10 years of menopause onset—or in women under age 60—demonstrates a different safety and efficacy profile than therapy started in older women further from menopause.
Younger women starting hormone therapy closer to menopause show:
- Reduced cardiovascular events: Some studies indicate reduced heart disease risk when estrogen is initiated early, contrasting with increased risk when started later
- Lower all-cause mortality: Women under 60 or within 10 years of menopause show reduced overall mortality with hormone therapy in multiple analyses
- Improved metabolic markers: Better lipid profiles, reduced visceral adiposity, and improved insulin sensitivity
- Maintained bone density: Significant reduction in fracture risk, particularly vertebral and hip fractures
The mechanistic explanation involves vascular health. Before significant atherosclerotic plaque develops, estrogen appears to have protective vascular effects—improving endothelial function, reducing LDL cholesterol, and limiting plaque formation. But once substantial plaque exists, estrogen may destabilize it, increasing rupture risk and cardiovascular events.
This isn’t just theoretical. A 2016 follow-up analysis of WHI participants found that women aged 50-59 who took estrogen alone had significantly reduced coronary artery calcium—a marker of atherosclerosis—compared to placebo. The effect was substantial: a 30-60% reduction depending on adherence.
Cardiovascular Benefits in Younger Women
The cardiovascular data in younger menopausal women has become increasingly compelling. A 2015 Cochrane review analyzing 19 trials found that hormone therapy started before age 60 or within 10 years of menopause was associated with reduced cardiovascular mortality and reduced all-cause mortality, with no increase in cardiovascular disease events.
More recent analyses confirm these findings. The overall mortality benefit appears driven primarily by cardiovascular protection in this younger group, though the mechanisms extend beyond simple cholesterol reduction.
Estrogen influences multiple cardiovascular risk pathways:
Lipid metabolism: Estrogen therapy typically reduces LDL cholesterol by 10-15% while increasing HDL by 10-15%. The specific changes depend on the type and route of estrogen administration, with transdermal estrogen showing more favorable effects on triglycerides.
Vascular function: Estrogen improves endothelial function—the ability of blood vessels to dilate properly—through increased nitric oxide production. This effect appears most beneficial before significant vascular disease develops.
Inflammation: While estrogen can increase some inflammatory markers, it appears to reduce vascular inflammation in healthy arteries, potentially slowing atherosclerosis progression.
Body composition: Menopausal hormone therapy helps maintain more favorable fat distribution, reducing accumulation of visceral adipose tissue that drives metabolic dysfunction and cardiovascular risk.
The Breast Cancer Question
Breast cancer risk remains the concern most frequently raised by patients considering hormone therapy. The data here requires careful interpretation.
The WHI found that combined estrogen-progestin therapy increased breast cancer risk—about 8 additional cases per 10,000 women per year. But estrogen alone in women who had undergone hysterectomy showed either no increased risk or possibly reduced risk, depending on the analysis.
This difference points to progestin as the likely driver of increased breast cancer risk in combined therapy. Multiple studies have since confirmed that different progestins carry different risks, with some showing lower breast cancer associations than others.
Progesterone—bioidentical to the hormone the body produces—may carry lower breast cancer risk than synthetic progestins, though definitive data remains limited. The EPIC cohort study found no increased breast cancer risk with estrogen plus progesterone, contrasting with increased risk from synthetic progestins.
For women with intact uteruses who require progestin to protect the endometrium, the choice of progestin and possibly delivery method (oral vs. intrauterine) may influence breast cancer risk. These are conversations worth having with knowledgeable clinicians.
Metabolic Effects and Weight Management
One benefit of menopausal hormone therapy that doesn’t receive adequate attention involves metabolic health and body composition. The menopausal transition typically brings unfavorable changes: increased visceral fat, reduced lean mass, declining metabolic rate, and worsening insulin sensitivity.
Hormone therapy mitigates many of these changes. Multiple studies show that estrogen therapy:
- Reduces visceral adipose tissue accumulation
- Helps maintain lean body mass
- Improves insulin sensitivity
- Reduces development of metabolic syndrome
- Lowers risk of type 2 diabetes
These metabolic benefits likely contribute to cardiovascular protection and overall mortality reduction seen in younger women initiating therapy.
Emerging research suggests that combining hormone therapy with other metabolic interventions may enhance benefits. The combination of estrogen therapy with GLP-1 receptor agonists like tirzepatide or semaglutide represents a particularly intriguing possibility for managing the metabolic changes of menopause, though clinical data specifically examining this combination remains limited.
The rationale is sound: estrogen addresses hormonal drivers of metabolic change, while GLP-1 agonists improve glucose control, reduce appetite, and promote weight loss. For women struggling with weight gain and metabolic dysfunction during the menopausal transition, this combination approach may prove more effective than either intervention alone.
Bone Health Benefits
One of the most established benefits of hormone therapy involves bone health. Estrogen deficiency accelerates bone loss, and hormone replacement effectively prevents this decline.
The WHI demonstrated substantial fracture reduction with hormone therapy: 24% reduction in hip fractures and 34% reduction in vertebral fractures. These benefits appeared consistent across age groups and bone density levels.
For women at increased fracture risk—those with low bone density, strong family history, or other risk factors—hormone therapy represents an evidence-based prevention strategy, particularly if initiated at menopause onset when bone loss typically accelerates.
The bone benefits persist during treatment but diminish after discontinuation, suggesting that duration of therapy should be individualized based on fracture risk and other considerations.
Quality of Life and Symptom Management
The FDA warnings focused primarily on chronic disease risks, potentially overshadowing what remains hormone therapy’s strongest indication: management of vasomotor symptoms.
For women experiencing severe hot flashes and night sweats that impair sleep, work performance, and quality of life, hormone therapy remains the most effective treatment available. Nothing else—not SSRIs, not gabapentin, not herbal supplements—matches estrogen’s efficacy for these symptoms.
Sleep disruption from night sweats carries its own health consequences, including increased cardiovascular risk, metabolic dysfunction, and cognitive impairment. Treating severe vasomotor symptoms may therefore provide health benefits beyond just symptom relief.
Hormone therapy also effectively treats genitourinary syndrome of menopause—vaginal dryness, painful intercourse, and urinary symptoms that affect roughly half of postmenopausal women. Low-dose vaginal estrogen provides benefits with minimal systemic absorption and no increased risk of the complications associated with oral hormone therapy.
Individual Risk Assessment
The shift in FDA guidance reflects growing recognition that hormone therapy decisions require individualized risk-benefit analysis, not blanket recommendations.
Factors that influence this calculation include:
Age and time since menopause: The most critical variable. Women under 60 or within 10 years of menopause show the most favorable risk-benefit profiles.
Personal and family history: Breast cancer history, cardiovascular disease history, blood clot history, and liver disease all influence recommendations.
Symptom severity: The benefit calculation differs dramatically for women with severe, life-disrupting symptoms versus those considering therapy primarily for disease prevention.
Bone density and fracture risk: Women at higher fracture risk gain greater benefit from hormone therapy’s bone protection.
Individual preferences and values: Different women reasonably weigh risks and benefits differently based on their priorities and concerns.
Formulation and Route Considerations
Not all hormone therapy formulations carry equivalent risks. Evidence suggests that:
- Transdermal estrogen (patches, gels) may carry lower blood clot risk than oral estrogen
- Micronized progesterone may carry lower breast cancer and cardiovascular risk than some synthetic progestins
- Lower doses generally carry lower risks while still providing benefits for many women
- Vaginal estrogen for genitourinary symptoms has minimal systemic absorption and extremely low risk
These distinctions matter. A woman using low-dose transdermal estradiol with micronized progesterone has a different risk profile than one taking higher-dose oral conjugated equine estrogen with medroxyprogesterone acetate.
What the FDA Changes Mean
The anticipated revision of black box warnings doesn’t mean hormone therapy is risk-free or appropriate for everyone. It means we’ve learned that risks and benefits vary substantially based on patient characteristics and treatment timing.
For women in their 50s experiencing significant menopausal symptoms, the accumulating evidence suggests that short-to-moderate term hormone therapy carries a favorable risk-benefit profile. The blanket warnings that emerged from the 2002 WHI may have unnecessarily frightened this group away from an effective treatment.
For older women considering hormone therapy primarily for disease prevention rather than symptom management, the calculation differs. Here, the evidence suggests less benefit and potentially increased risk.
The key insight is that age and timing fundamentally alter the risk-benefit equation. A 52-year-old woman two years past menopause has a different calculus than a 65-year-old woman 15 years past menopause.
Practical Implications
What does this mean for women making decisions about hormone therapy?
First, if you’re experiencing significant menopausal symptoms and you’re under 60 or within 10 years of menopause, hormone therapy deserves consideration. The evidence doesn’t support avoiding treatment based on the generalized fears that arose from the original WHI interpretation.
Second, formulation and route matter. Discuss with your physician whether transdermal estrogen and micronized progesterone might be appropriate. These options may offer a more favorable risk profile than older formulations.
Third, regular reassessment is important. Hormone therapy isn’t necessarily lifelong treatment. Many women use it for several years to manage the acute symptoms of the menopausal transition, then taper off as symptoms naturally diminish.
Fourth, hormone therapy can be part of a comprehensive approach to menopausal health that includes exercise, nutrition, stress management, sleep optimization, and—when appropriate—other medications addressing specific concerns like bone health or metabolic function.
Finally, find a clinician knowledgeable about current hormone therapy evidence. Not all physicians have kept up with the evolving understanding of risks and benefits. A clinician well-versed in menopause medicine can help navigate the nuances and individualize recommendations.
Looking Forward
The anticipated FDA warning changes represent a course correction based on accumulated evidence. This is how medicine should work—initial findings prompt caution, then nuanced analysis reveals where that caution is most warranted and where it may have been overly broad.
For the generation of women who went through menopause in the shadow of the 2002 WHI findings, many may have avoided a treatment that could have significantly improved their quality of life with acceptable risk. That’s unfortunate, but it reflects the information available at the time and the appropriate precautionary response to concerning study results.
Going forward, we can apply what we’ve learned: hormone therapy decisions require individualization based on age, timing, symptoms, risk factors, and patient preferences. There’s no single right answer for all women, but there are increasingly clear frameworks for making informed decisions.
The revision of FDA warnings won’t eliminate all controversy or uncertainty. But it should help ensure that women and their physicians have access to current evidence rather than making decisions based on generalized warnings that don’t reflect individual circumstances.
For women navigating menopause, that’s a meaningful step forward.
Dr. Pradeep Albert is a regenerative medicine physician and musculoskeletal radiologist specializing in advanced cellular therapies and longevity science. He is the author of “Exosomes, PRP, and Stem Cells in Musculoskeletal Medicine” and co-author of “Lifespan Decoded: How to Hack Your Biology for a Longer, Healthier Life.”